A Review of Research on Aerosol Hygroscopic Growth in Severe Nuclear Reactor Accidents
-
摘要: 可溶性气溶胶(以下简称气溶胶)的吸湿增长是影响核反应堆严重事故中放射性产物动力学行为的关键因素之一,本文对吸湿增长的理论模型、实验测量方法以及核安全领域吸湿增长的研究进展进行了总结。核安全领域气溶胶吸湿增长理论模型以Köhler理论为基础,描述了吸湿增长过程中气溶胶热物性等参数与环境参数的关系,在此基础上发展的多种改进模型更适用于实际问题的分析,已在NAUA-HYGROS、MELCOR等核安全气溶胶计算程序中得到广泛应用。实验测量也是研究气溶胶吸湿增长特性的重要手段,相比于可测量气溶胶整体吸湿能力但结果较为粗糙的重量法,电力平衡法、HTDMA法精度较高,且具备实时测量单个颗粒及多模态颗粒群的吸湿能力,在核事故气溶胶吸湿增长特性实验研究方面具有潜在应用前景。本文最后总结了严重核事故领域吸湿增长的现有应用研究,包括核事故典型气溶胶吸湿增长特性的理论模型与数值计算应用研究、吸湿增长实验研究,数值计算研究表明,将气溶胶吸湿增长特性纳入到核事故气溶胶计算程序中可以实现对核事故发生后气溶胶行为更为准确的预测分析,相关实验得到了CsOH、CsI等典型核事故气溶胶的吸湿增长特性。Abstract: The hygroscopic growth of soluble aerosols (hereinafter referred to as aerosols) is one of the key factors affecting the dynamic behavior of radioactive products in serious nuclear reactor accidents. In this paper, the theoretical model of hygroscopic growth, experimental measurement scheme and research progress of hygroscopic growth in nuclear safety field are summarized. Based on Köhler theory, aerosol hygroscopic growth theoretical model in the field of nuclear safety describes the relationship between aerosol thermophysical parameters and environmental parameters in the process of hygroscopic growth. On this basis, a variety of improved models are more suitable for the analysis of practical problems, and have been widely used in nuclear safety aerosol calculation programs such as NAUA-HYGROS and MELCOR. Experimental measurement is also an important means to study the hygroscopic growth characteristics of aerosols. Compared with the gravimetric method, which can measure the overall hygroscopic ability of aerosols, but the results are relatively rough, the power balance method and the HTDMA method have higher accuracy, and can measure the hygroscopic ability of single particles and multimodal particle groups in real time, and have potential application prospects in the experimental study of the aerosol hygroscopic growth characteristics of nuclear accidents. At the end of this paper, the existing applied researches on hygroscopic growth in the field of serious nuclear accidents are summarized, including the theoretical model and numerical application of typical aerosol hygroscopic growth in nuclear accidents, and the experimental study of hygroscopic growth. Numerical studies show that incorporating aerosol hygroscopic growth characteristics into the aerosol calculation program of nuclear accident can achieve more accurate prediction and analysis of aerosol behavior after nuclear accident. The hygroscopic growth characteristics of CsOH, CSI and other typical nuclear accident aerosols were obtained by relevant experiments.
-
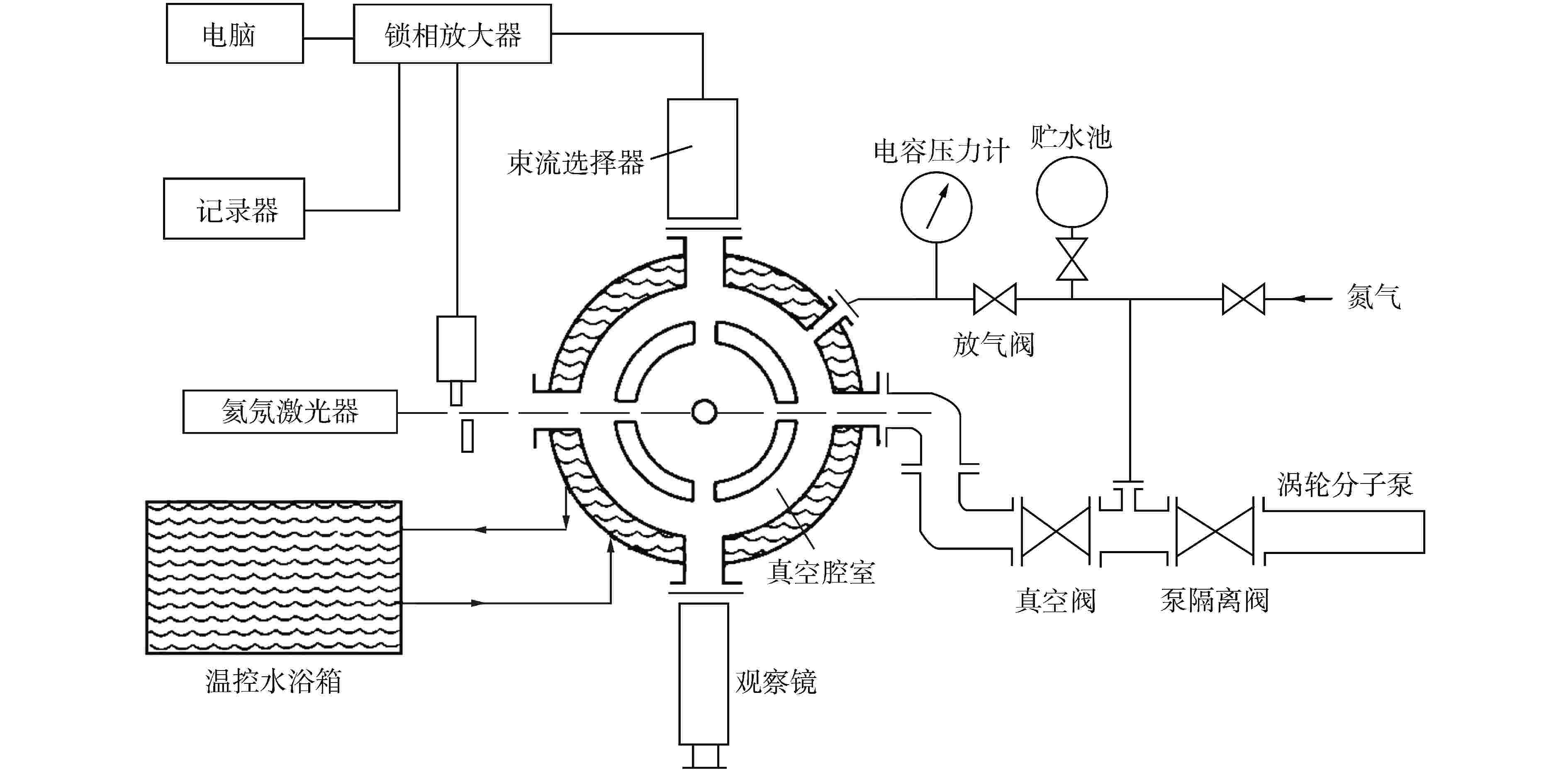
图 1 电力平衡装置示意图[56]
Figure 1. Schematic Diagram of Electric Power Balance Apparatus
图 2 HTDMA系统流程示意图[58]
Figure 2. Schematic Diagram of HTDMA System Flow
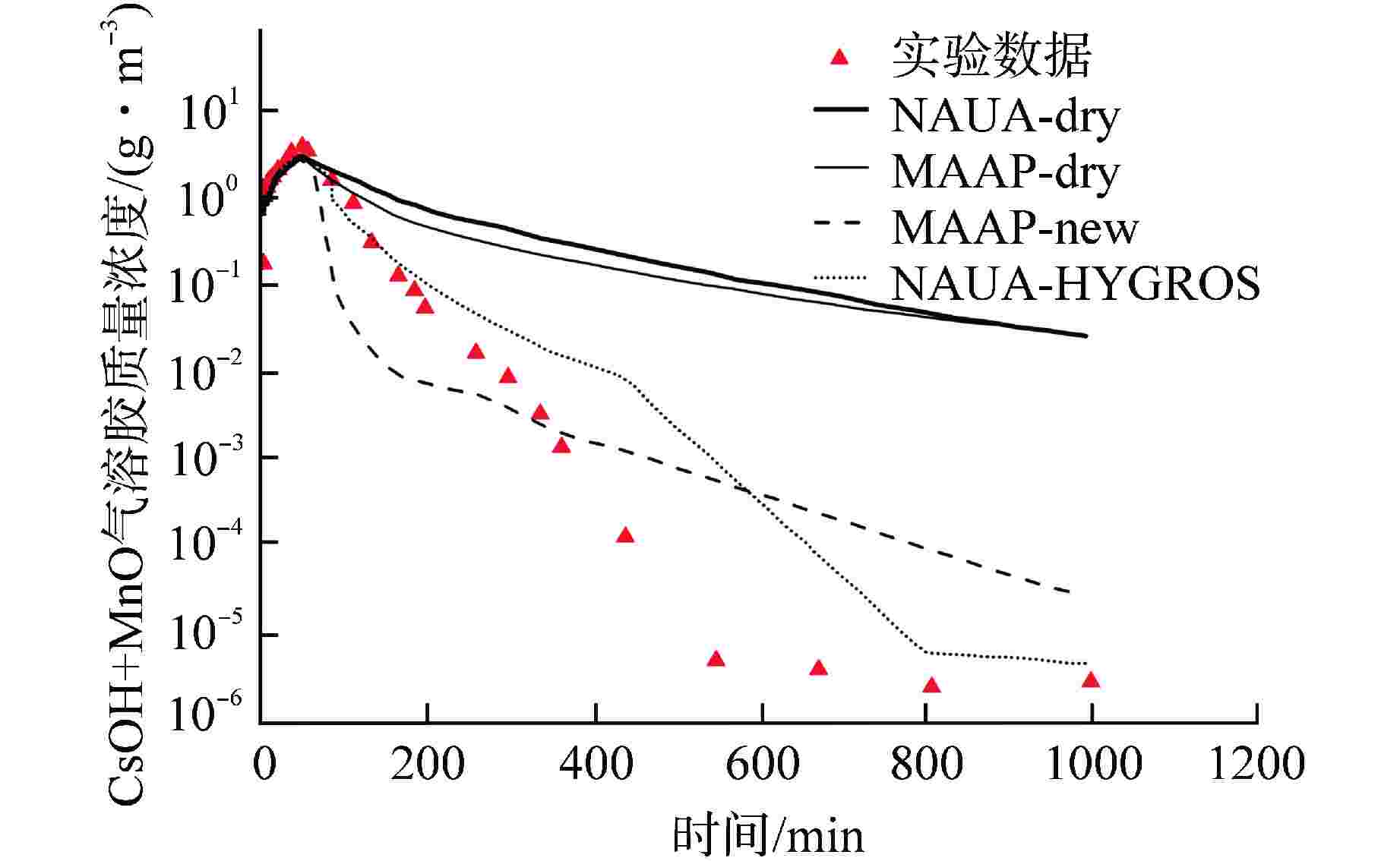
图 3 LACE实验中LA2工况(点)与原始及改进后的MAAP程序、NAUA及NAUA-HYGROS程序的计算结果(线)对比[44]
Figure 3. Comparison of Calculation Results (Lines) between LA2 Working Condition (Point) and Original and Improved MAAP Program, NAUA and NAUA-HYGROS Program in LACE Experiment
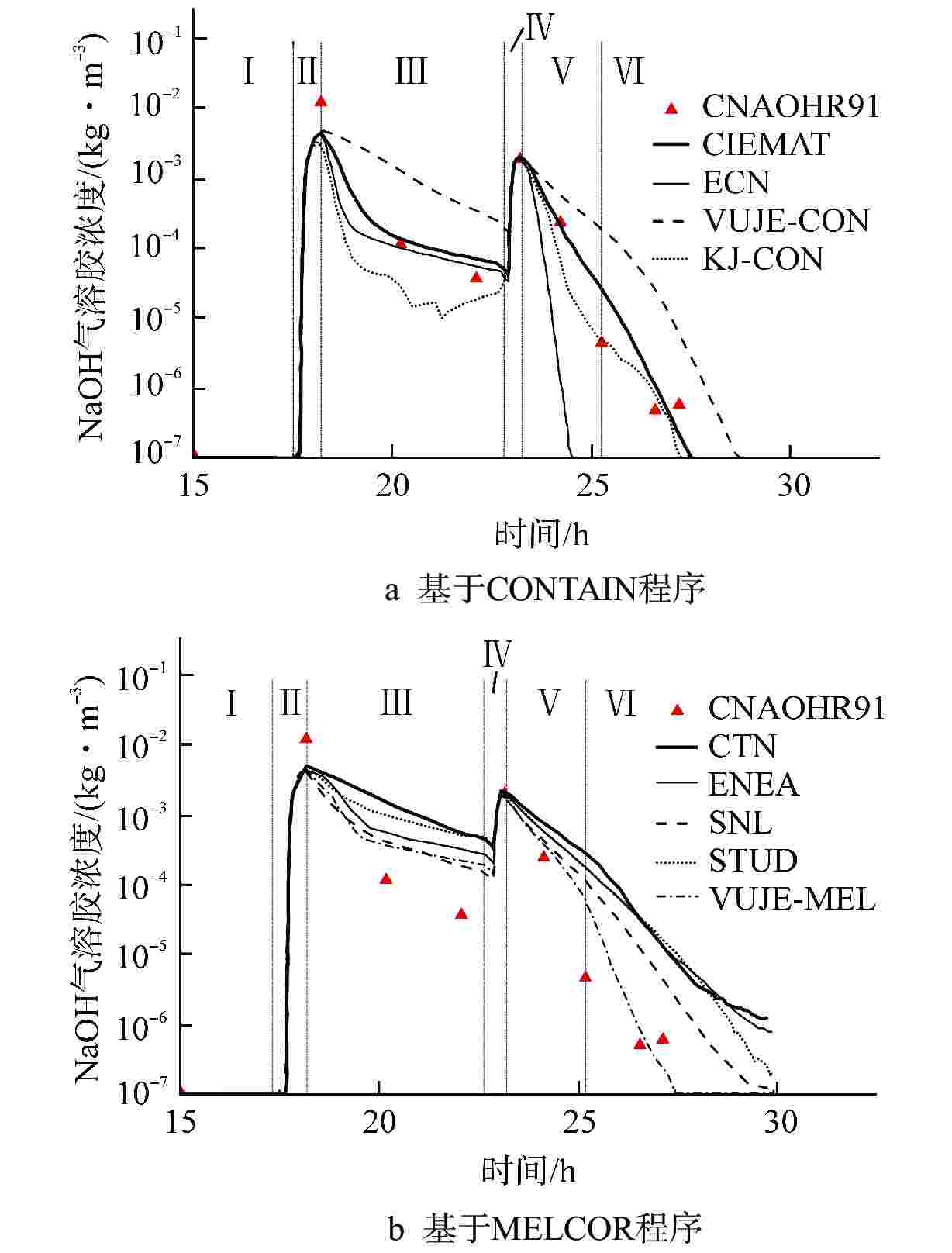
图 4 VANAM实验CNAOHR91工况与基于CONTAIN程序及MELCOR程序的计算结果对比[69]
CITMAT、ECN、VUJE-CON、KI-CON、CTN、ENEA、SNL、STUD、VUJE-MEL——不同研究机构的项目代号;图中罗马数字表示不同的阶段
Figure 4. Comparison of CNAOHR91 Working Condition in VANAM Experiment with the Calculation Results based on the CONTAIN Program and the MELCOR Program
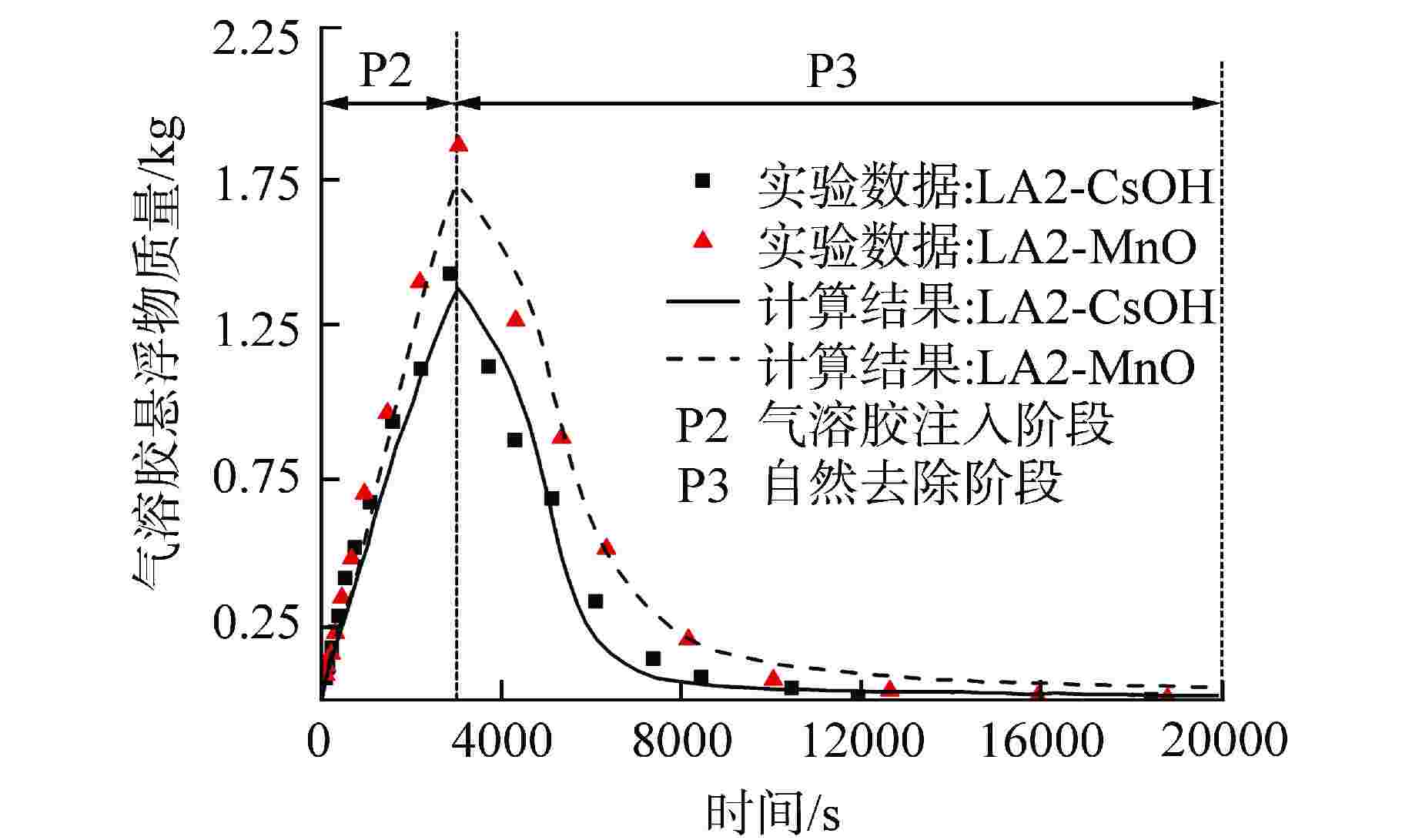
图 5 实验空间内CsOH或MnO气溶胶气溶胶悬浮物质量随时间变化:ATHROC模型(线)与LACE实验LA2工况(点)[34]
Figure 5. Variation of Aerosol Suspension Mass of CsOH or MnO Aerosols with Time in Experimental Space: ATHROC Model (Line) and LACE Experiment LA2 Working Condition (Point)
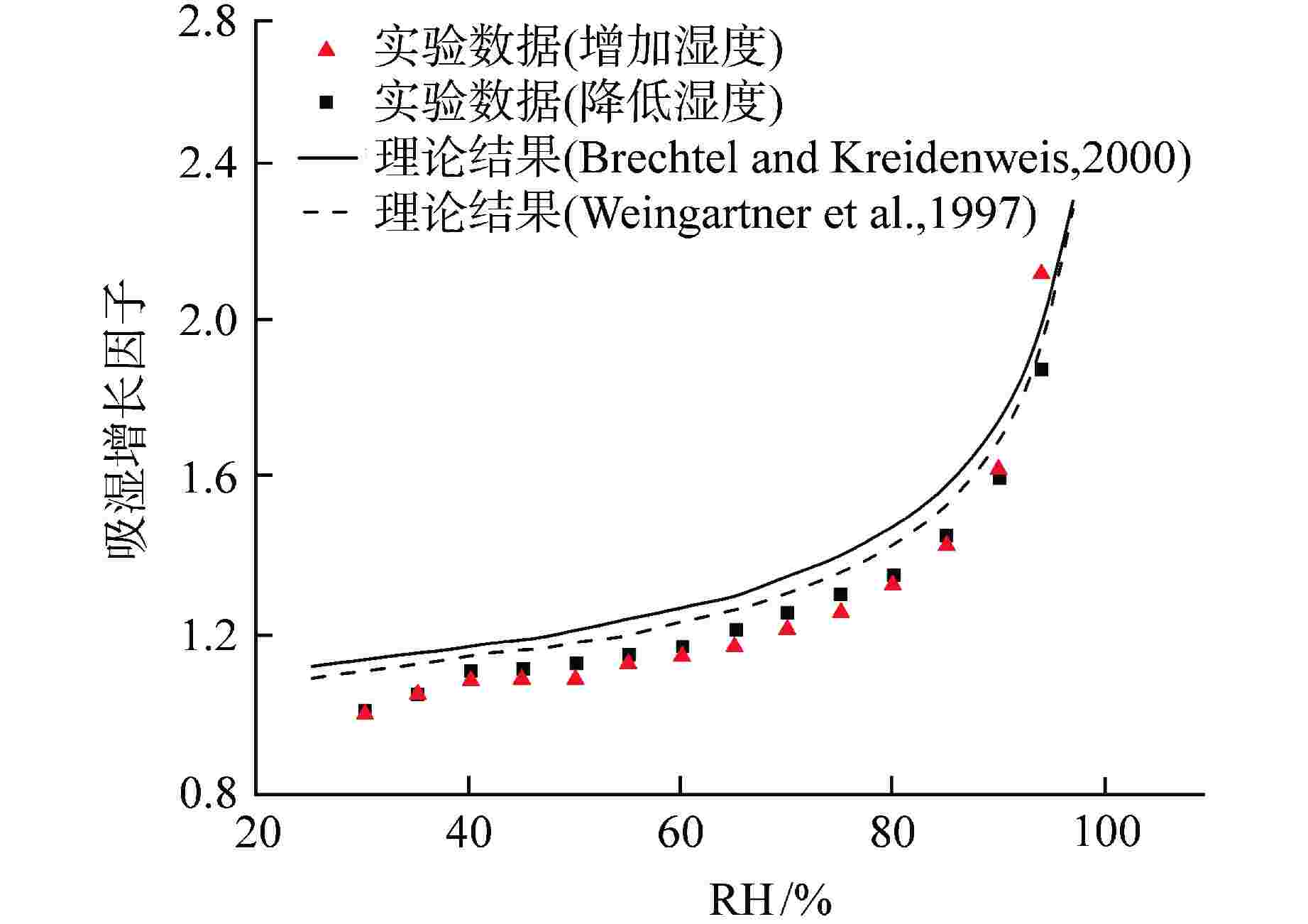
图 6 0.1 μm CsI颗粒吸湿增长因子实验及理论结果[12]
Figure 6. Experimental and Theoretical Hygroscopic Growth Factors of 0.1 μm CsI Particle
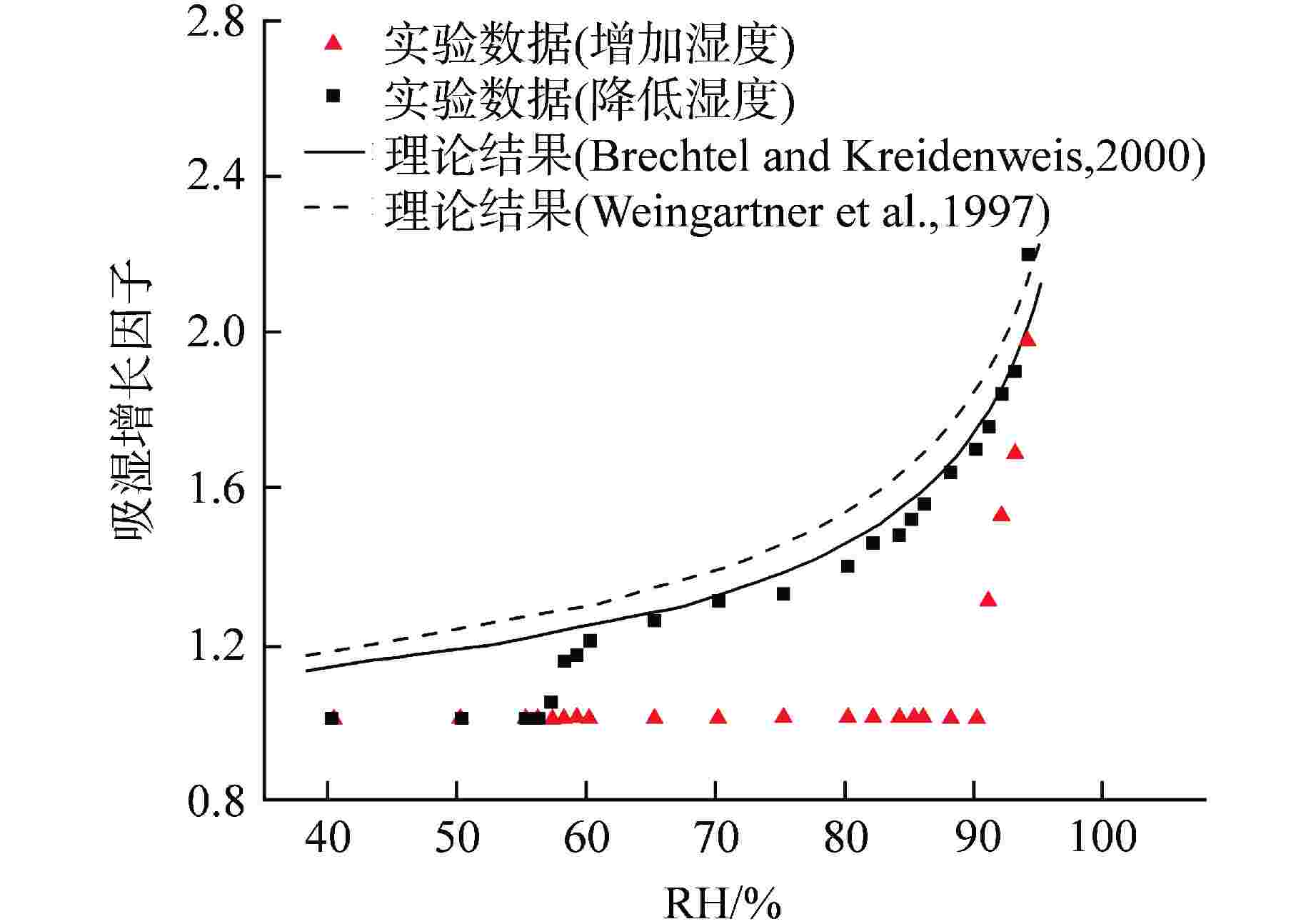
图 7 0.1 μm CsOH颗粒吸湿增长因子实验及理论结果[12]
Figure 7. Experimental and Theoretical Hygroscopic Growth Factors of 0.1 μm CsOH Particle
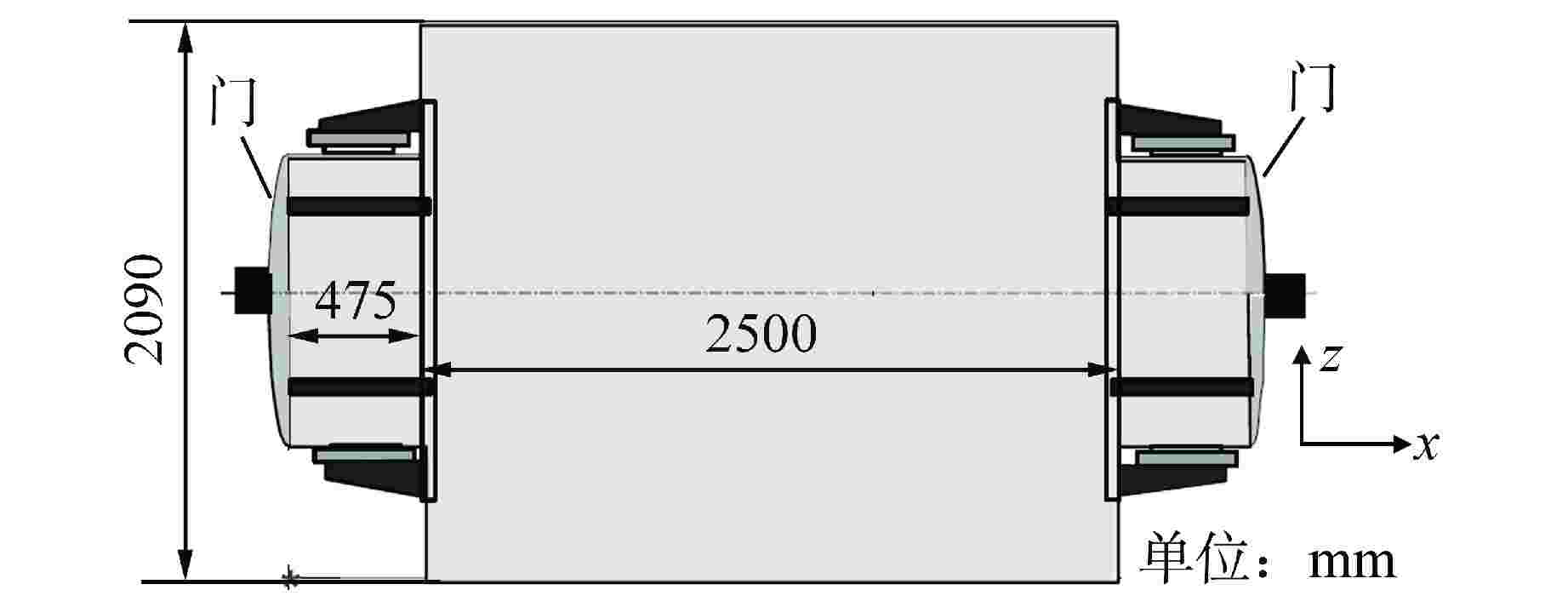
图 8 KAEVER实验罐体俯视图[76]
Figure 8. Top View of KAEVER Experimental Tank
-
[1] 李应治,周艳民,孙中宁,等. 鼓泡过滤可溶性气溶胶特性实验研究[J]. 核动力工程,2019, 40(S2): 21-25. [2] 杨林民,周涛,陆道纲. 压水堆严重事故下气溶胶热泳沉积规律[J]. 原子能科学技术,2008, 42(1): 63-66. [3] SUN H P, DENG J, ZHU D H, et al. Development of a new method for simulating gas-to-particle conversion during sodium pool fires in SFR containment[J]. Nuclear Technology, 2020, 206(10): 1481-1493. [4] 孙雪霆,陈林林,魏严凇,等. 非能动安全壳冷却对严重事故下气溶胶沉积影响分析[J]. 原子能科学技术,2016, 50(12): 2219-2223. [5] 佟立丽,曹学武. 非能动安全壳冷却机制安全壳大气净化分析[J]. 科技导报,2012, 30(20): 29-32. [6] 陈巧艳,杨志义,周涛,等. 百万千瓦级压水堆核电厂二级PSA源项分析与研究[J]. 核动力工程,2016, 37(02): 97-101. [7] 杨志义,陈鹏,种毅敏,等. 大型干式安全壳严重事故下超压失效概率研究[J]. 核科学与工程,2016, 36(3): 380-386. [8] 宫厚军,杨星团,黄彦平,等. 倾斜条件下一体化反应堆模拟回路单相自然循环实验与数值研究[J]. 核动力工程,2014, 35(05): 89-93. [9] 熊万玉,宫厚军,郗昭,等. RELAP5程序应用于二次侧非能动余热排出系统设计的初步评价[J]. 核动力工程,2015, 36(2): 143-146. [10] COVERT D S, CHARLSON R J, AHLQUIST N C. A Study of the relationship of chemical composition and humidity to light scattering by aerosols[J]. Journal of Applied Meteorology, 1972, 11(6): 968-976. [11] SOFFER L, BURSON S B, FERRELL C M, et al. Accident source terms for light-water nuclear power plants: NUREG-1465[R]. Washington: Nuclear Regulatory Commission, 1995. [12] MISHRA G, MANDARIYA A K, TRIPATHI S N, et al. Hygroscopic growth of CsI and CsOH particles in context of nuclear reactor accident research[J]. Journal of Aerosol Science, 2019, 132: 60-69. [13] COHEN M D, FLAGAN R C, SEINFELD J H. Studies of concentrated electrolyte solutions using the electrodynamic balance. 1. Water activities for single-electrolyte solutions[J]. The Journal of Physical Chemistry, 1987, 91(17): 4563-4574. [14] TANG I N. Phase transformation and growth of aerosol particles composed of mixed salts[J]. Journal of Aerosol Science, 1976, 7(5): 361-371. doi: 10.1016/0021-8502(76)90022-7 [15] GYSEL M, WEINGARTNER E, BALTENSPERGER U. Hygroscopicity of aerosol particles at low temperatures. 2. Theoretical and experimental hygroscopic properties of laboratory generated aerosols[J]. Environmental Science & Technology, 2002, 36(1): 63-68. [16] PENG C G, CHAN M N, CHAN C K. The hygroscopic properties of dicarboxylic and multifunctional acids: measurements and UNIFAC predictions[J]. Environmental Science & Technology, 2001, 35(22): 4495-4501. [17] BADGER C L, GEORGE I, GRIFFITHS P T, et al. Phase transitions and hygroscopic growth of aerosol particles containing humic acid and mixtures of humic acid and ammonium sulphate[J]. Atmospheric Chemistry and Physics, 2006, 6(3): 755-768. [18] NOLL K E, MUELLER P K, IMADA M. Visibility and aerosol concentration in urban air[J]. Atmospheric Environment, 1968, 2(5): 465-475. [19] KASTEN F. Visibility forecast in the phase of pre-condensation[J]. Tellus, 1969, 21(5): 631-635. [20] KOTCHENRUTHER R A, HOBBS P V. Humidification factors of aerosols from biomass burning in Brazil[J]. Journal of Geophysical Research:Atmospheres, 1998, 103(D24): 32081-32089. [21] KöHLER H. The nucleus in and the growth of hygroscopic droplets[J]. Transactions of the Faraday Society, 1936, 32: 1152-1161. [22] PRUPPACHER H R, KLETT J D. Microphysics of clouds and precipitation[M]. Dordrecht, Boston: Kluwer Academic Publishers, 1997:172-175 [23] SHULMAN M L, JACOBSON M C, CARLSON R J, et al. Dissolution behavior and surface tension effects of organic compounds in nucleating cloud droplets[J]. Geophysical Research Letters, 1996, 23(3): 277-280. [24] FACCHINI M C, DECESARI S, MIRCEA M, et al. Surface tension of atmospheric wet aerosol and cloud/fog droplets in relation to their organic carbon content and chemical composition[J]. Atmospheric Environment, 2000, 34(28): 4853-4857. [25] WEINGARTNER E, BURTSCHER H, BALTENSPERGER U. Hygroscopic properties of carbon and diesel soot particles[J]. Atmospheric Environment, 1997, 31(15): 2311-2327. [26] BRECHTEL F J, KREIDENWEIS S M. Predicting particle critical supersaturation from hygroscopic growth measurements in the humidified TDMA. Part I: theory and sensitivity studies[J]. Journal of the Atmospheric Sciences, 2000, 57(12): 1854-1871. [27] GREGOR H P. Electrolyte solutions [J]. Journal of Applied Polymer Science, 1960, 3(8): 255. [28] CLEGG S L, PITZER K S. Thermodynamics of multicomponent, miscible, ionic solutions: generalized equations for symmetrical electrolytes[J]. The Journal of Physical Chemistry, 1992, 96(8): 3513-3520. [29] PITZER K S. Thermodynamics of electrolytes. I. Theoretical basis and general equations[J]. The Journal of Physical Chemistry, 1973, 77(2): 268-277. [30] PITZER K S, MAYORGA G. Thermodynamics of electrolytes. II. Activity and osmotic coefficients for strong electrolytes with one or both ions univalent[J]. The Journal of Physical Chemistry, 1973, 77(19): 2300-2308. [31] PITZER K S. Activity coefficients in electrolyte solutions[M]. 2nd ed. Boca Raton: CRC Press, 2018:98-100 [32] LU J J, ZHANG T Q, MAO Y W, et al. An improved sectional model to simulate multi-component aerosol dynamics in a containment of pressurized water reactor[J]. Journal of Aerosol Science, 2021, 157: 105800. [33] LI Y Z, ZHOU Y M, SUN Z N, et al. Analysis of hygroscopic growth properties of soluble aerosol under severe nuclear accidents conditions[J]. Progress in Nuclear Energy, 2020, 127: 103464. [34] SUN H P, DENG J, ZHANG Y P, et al. Development and validation of a new module in the ATHROC for simulation of aerosol particles removal by spray in containment[J]. Nuclear Engineering and Design, 2020, 364: 110630. doi: 10.1016/j.nucengdes.2020.110630 [35] LIU B Y H, PUI D Y H, WHITBY K T, et al. The aerosol mobility chromatograph: a new detector for sulfuric acid aerosols[J]. Atmospheric Environment, 1978, 12(1-3): 99-104. [36] DECARLO P F, SLOWIK J G, WORSNOP D R, et al. Particle morphology and density characterization by combined mobility and aerodynamic diameter measurements. Part 1: theory[J]. Aerosol Science and Technology, 2004, 38(12): 1185-1205. doi: 10.1080/027868290903907 [37] BISKOS G, MALINOWSKI A, RUSSELL L M, et al. Nanosize effect on the deliquescence and the efflorescence of sodium chloride particles[J]. Aerosol Science and Technology, 2006, 40(2): 97-106. [38] WAGNER P E. Aerosol growth by condensation[M]. Berlin, Heidelberg: Springer, 1982: 129-178. [39] CHANG R, DAVIS E J. Interfacial conditions and evaporation rates of a liquid droplet[J]. Journal of Colloid and Interface Science, 1974, 47(1): 65-76. doi: 10.1016/0021-9797(74)90080-0 [40] FUCHS N A, SUTUGIN A G. High-dispersed aerosols[M].Oxford: Pergamon, 1971: 1. [41] KULMALA M, MAJEROWICZ A, WAGNER P E. Condensational growth at large vapour concentration: limits of applicability of the mason equation[J]. Journal of Aerosol Science, 1989, 20(8): 1023-1026. [42] JOKINIEMI J. The growth of hygroscopic particles during severe core melt accidents[J]. Nuclear Technology, 1988, 83(1): 16-23. [43] [44] FYNBO P B, HäGGBLOM H, JOKINIEMI J. Aerosol transport in severe reactor accidents. Final report of the NKA project AKTI-160[M]. Nyköping: Nordiska Kontaktorganet för Atomenergifrågor, 1990. [45] JOKINIEMI J. Effect of selected binary and mixed solutions on steam condensation and aerosol behavior in containment[J]. Aerosol Science and Technology, 1990, 12(4): 891-902. [46] HUMPHRIES L L, FIGUEROA V G, YOUNG M F, et al. MELCOR computer code manuals volume 1: primer and users' guide: SAND-2015-6691R[R]. Albuquerque: Sandia National Laboratory, 2015. [47] CHARLSON R J. Atmospheric visibility related to aerosol mass concentration: review[J]. Environmental Science & Technology, 1969, 3(10): 913-918. [48] ZHAO Y F, WANG X, CAI Y J, et al. Measurements of atmospheric aerosol hygroscopic growth based on multi-channel Raman-Mie lidar[J]. Atmospheric Environment, 2021, 246: 118076. [49] ZHANG Q N, ZHAO L J, CHEN S H, et al. Hygroscopic property of inorganic salts in atmospheric aerosols measured with physisorption analyzer[J]. Atmospheric Environment, 2021, 247: 118171. [50] ZHAO P S, DU X, SU J, et al. Aerosol hygroscopicity based on size-resolved chemical compositions in Beijing[J]. Science of the Total Environment, 2020, 716: 137074. [51] 刘强,王明星,李晶,等. 大气气溶胶研究现状和发展趋势[J]. 中国粉体技术,1999, 5(3): 17-23. [52] 王轩,陈建华,王玮. 气溶胶吸湿特性研究现状[J]. 中国粉体技术,2010, 16(1): 101-107. [53] 王轩. 气溶胶吸湿特性研究[D]. 北京: 中国环境科学研究院, 2010. [54] WINKLER P. The growth of atmospheric aerosol particles as a function of the relative humidity—II. An improved concept of mixed nuclei[J]. Journal of Aerosol Science, 1973, 4(5): 373-387. [55] TANG I N, MUNKELWITZ H R, WANG N. Water activity measurements with single suspended droplets: the NaCl-H2O and KCl-H2O systems[J]. Journal of Colloid and Interface Science, 1986, 114(2): 409-415. doi: 10.1016/0021-9797(86)90426-1 [56] TANG I N, MUNKELWITZ H R. Water activities, densities, and refractive indices of aqueous sulfates and sodium nitrate droplets of atmospheric importance[J]. Journal of Geophysical Research:Atmospheres, 1994, 99(D9): 18801-18808. [57] 刘新罡,张远航. 大气气溶胶吸湿性质国内外研究进展[J]. 气候与环境研究,2010, 15(6): 808-816. [58] 吴奕霄. 大气气溶胶的吸湿性观测研究——以南京和黄山为例[D]. 南京: 南京信息工程大学, 2014. [59] CARRICO C M, ROOD M J, OGREN J A, et al. Aerosol Optical properties at Sagres, Portugal during ACE-2[J]. Tellus B, 2000, 52(2): 694-715. [60] HANSSON H C, ROOD M J, KOLOUTSOU-VAKAKIS S, et al. NaCl aerosol particle hygroscopicity dependence on mixing with organic compounds[J]. Journal of Atmospheric Chemistry, 1998, 31(3): 321-346. [61] WEIS D D, EWING G E. Infrared spectroscopic signatures of (NH4)2SO4 aerosols[J]. Journal of Geophysical Research:Atmospheres, 1996, 101(D13): 18709-18720. [62] HOSEYNI M S, WASSEL A T. Growth of aerosol particles in a steam environment and its effect on removal[J]. Nuclear Engineering and Design, 1986, 97(1): 103-109. [63] OWCZARSKI P C, BURK K W. SPARC-90: a code for calculating fission product capture in suppression pools: MUREG/CR-5765[R]. Richland: Pacific Northwest Laboratory, 1991. [64] 李应治, 马钎朝, 刁寒, 等. 利用Köhler理论分析碘化铯和氢氧化铯的吸湿增长特性[C]//第十六届全国反应堆热工流体学术会议暨中核核反应堆热工水力技术重点实验室2019年学术年会论文集. 惠州: 中国核学会核能动力分会, 2019: 565-572 [65] ZHANG C Y, XING Y B, TAO D P. Prediction of activity and osmotic coefficients of fission product systems CsOH + CsX (X = Cl, Br, I) at 298.15 K[J]. Journal of Radioanalytical and Nuclear Chemistry, 2020, 323(2): 773-784. [66] BUNZ H, KOYRO M, SCHOECK W. NAUA Mod 4. A code for calculating aerosol behaviour in LWR core melt accidents. Code description and users manual: KFK-3554[R]. German: Laboratorium für Aerosolphysik und Filtertechnik, 1983 [67] HILLIARD R K, MUHLESTEIN L D, ALBIOL T J. Final report of experimental results of LACE test LA2-Failure to isolate containment: LA-TR-007[R]. Richland: Westinghouse Hanford Company, 1987. [68] KANZLEITER T, FISCHER K O, ALLELEIN H J, et al. The VANAM experiments M1 and M2 - test results and multi-compartmental analysis[J]. Journal of Aerosol Science, 1991, 22: 697-700. [69] FISCHER K, KANZLEITER T. Experiments and computational models for aerosol behaviour in the containment[J]. Nuclear Engineering and Design, 1999, 191(1): 53-67. [70] SONG Y M, KIM H D, PARK Y M, et al. Source term model accuracy evaluation in MELCOR code using international standard problem No. 44[J]. Journal of Nuclear Science and Technology, 2004, 41(S4): 439-442. [71] CHEN Y Z, WU Y W, WANG M J, et al. Development of a multi-compartment containment code for advanced PWR plant[J]. Nuclear Engineering and Design, 2018, 334: 75-89. [72] SCHMITT R C, BINGHAM G E, NORBERG J A. Simulated design basis accident tests of the Carolinas Virginia tube reactor containment: IN-1403[R]. Idaho Falls: Idaho Nuclear Corporation, 1970. [73] HILLIARD R K, COLEMAN L F, LINDEROTH C E, et al. Removal of iodine and particles from containment atmospheres by sprays: containment systems experiment interim report: BNWL-1244[R]. Washington: Pacific Northwest Laboratory, 1970. [74] JOKINIEMI J, KOISTINEN K, RAUNEMAA T. Experimental verification of hygroscopic aerosol growth in reactor accident conditions[J]. Nuclear Technology, 1990, 90(3): 394-407. doi: 10.13182/NT90-A34403 [75] SCHEIBEL H G, POSS G, WEBER D. K A E V E R: an experiment for an improved understanding of aerosol depletion processes in a reactor containment[J]. Journal of Aerosol Science, 1992, 23 : 209-212. [76] FIRNHABER M, FISCHER K, SCHWARZ S, et al. International standard problem ISP44 - KAEVER - experiments on the behavior of core-melt aerosols in a LWR containment: NEA/CSNI/R(2003)5[R]. France: Nuclear Energy Agency of the OECD (NEA), 2002 [77] FIRNHABER M, KANZLEITER T F, SCHWARZ S, et al. International standard problem ISP37: VANAM M3 - a multi compartment aerosol depletion test with hygroscopic aerosol material: NEA/CSNI/R(96)26[R]. France: Nuclear Energy Agency of the OECD (NEA), 1996 [78] LI Y Z, MA Q C, SUN Z N, et al. Study on calculation method of soluble aerosol removal efficiency under high humidity condition[J]. Frontiers in Energy Research, 2019, 7: 26. [79] 卢俊晶,张天琦,杨小明,等. 严重事故下吸湿性气溶胶的自然去除研究[J]. 核动力工程,2020, 41(1): 145-149. -




 下载:
下载: