Comparative Study on Corrosion Resistance of TaSiCN, TaAlSiCN and TaCrSiCN Coatings in NaCl-KCl Molten Salt
-
摘要: 为探究Al、Cr元素掺杂的TaSiCN涂层在NaCl-KCl熔盐中的耐腐蚀性能,利用射频磁控溅射技术并通过调控Ar和N2混合气体的比例在Ta合金基体上制备了近似N原子百分比的TaSiCN、TaAlSiCN和TaCrSiCN纳米复合涂层。系统研究了涂层的微观结构、化学组成和耐NaCl-KCl熔盐的腐蚀性能。结果表明:所有沉积态的涂层均由TaN、TaC和Ta(C,N)纳米晶相以及SiCx、SiNx、CNx和sp2无定型态碳等非晶相组成,其中掺杂元素Al、Cr也以非晶的形式存在于涂层之中。腐蚀后的涂层表面主要由Ta(C,N)、Ta2O5、Na2Ta4O11晶相成分和一些非晶相成分构成。涂层在NaCl-KCl熔盐中的腐蚀行为主要以氧化腐蚀为主,熔盐侵蚀为辅。通过对比发现,涂层耐腐蚀性顺序为:TaAlSiCN>TaSiCN>TaCrSiCN。本研究可为熔盐电解坩埚表面涂层技术的开发提供一种具有应用潜力的候选涂层材料。
-
关键词:
- TaSiCN涂层 /
- 磁控溅射技术 /
- Ta合金 /
- NaCl-KCl熔盐 /
- 抗腐蚀性
Abstract: In order to explore the corrosion resistance of Al and Cr doped TaSiCN coatings in NaCl-KCl molten salt, nano-composite coatings of TaSiCN, TaAlSiCN and TaCrSiCN with approximately N atomic percent were prepared on Ta alloy substrate by using RF magnetron sputtering technology and adjusting the ratio of Ar and N2 mixed gases. The microstructure, chemical composition and corrosion resistance of the coating in NaCl-KCl molten salt were systematically studied. The results show that all as-deposited coatings are composed of TaN, TaC and Ta(C,N) nanocrystals and amorphous carbon such as SiCx, SiNx, CNx and sp2, among which the doped Al and Cr elements also exist in the coatings in amorphous form. After corrosion, the coating surface is mainly composed of crystalline components such as Ta(C,N), Ta2O5 and Na2Ta4O11, and other amorphous components. The corrosion behavior of coating in NaCl-KCl molten salt is mainly oxidation corrosion, supplemented by molten salt corrosion. Through comparison, it is found that the order of corrosion resistance of three coatings is: TaAlSiCN > TaSiCN > TaCrSiCN. This study can provide a potential candidate coating material for the development of surface coating technology of molten salt electrolytic crucible.-
Key words:
- TaSiCN coating /
- Magnetron sputtering /
- Ta alloy /
- NaCl-KCl molten salt /
- Corrosion resistance
-
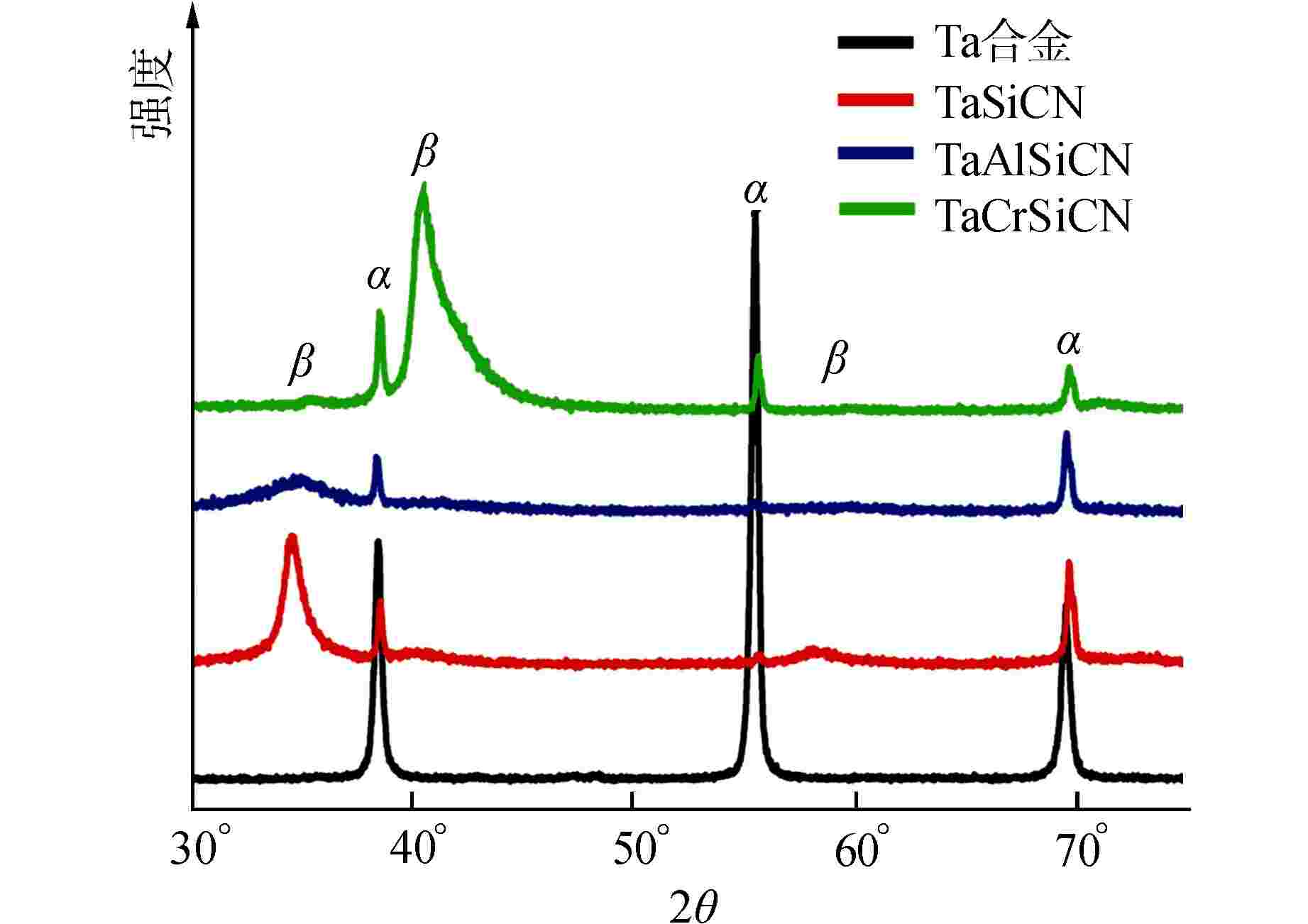
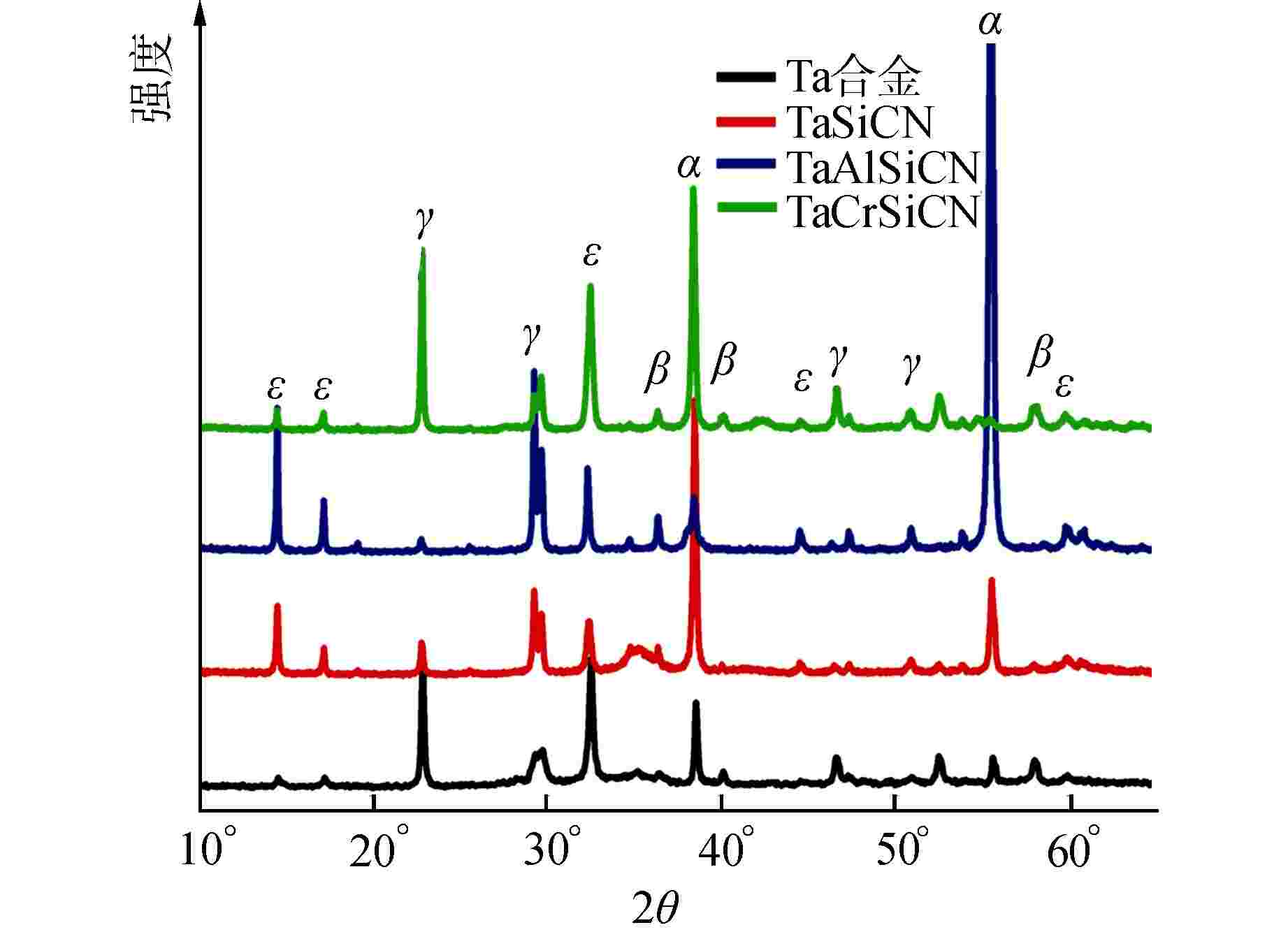
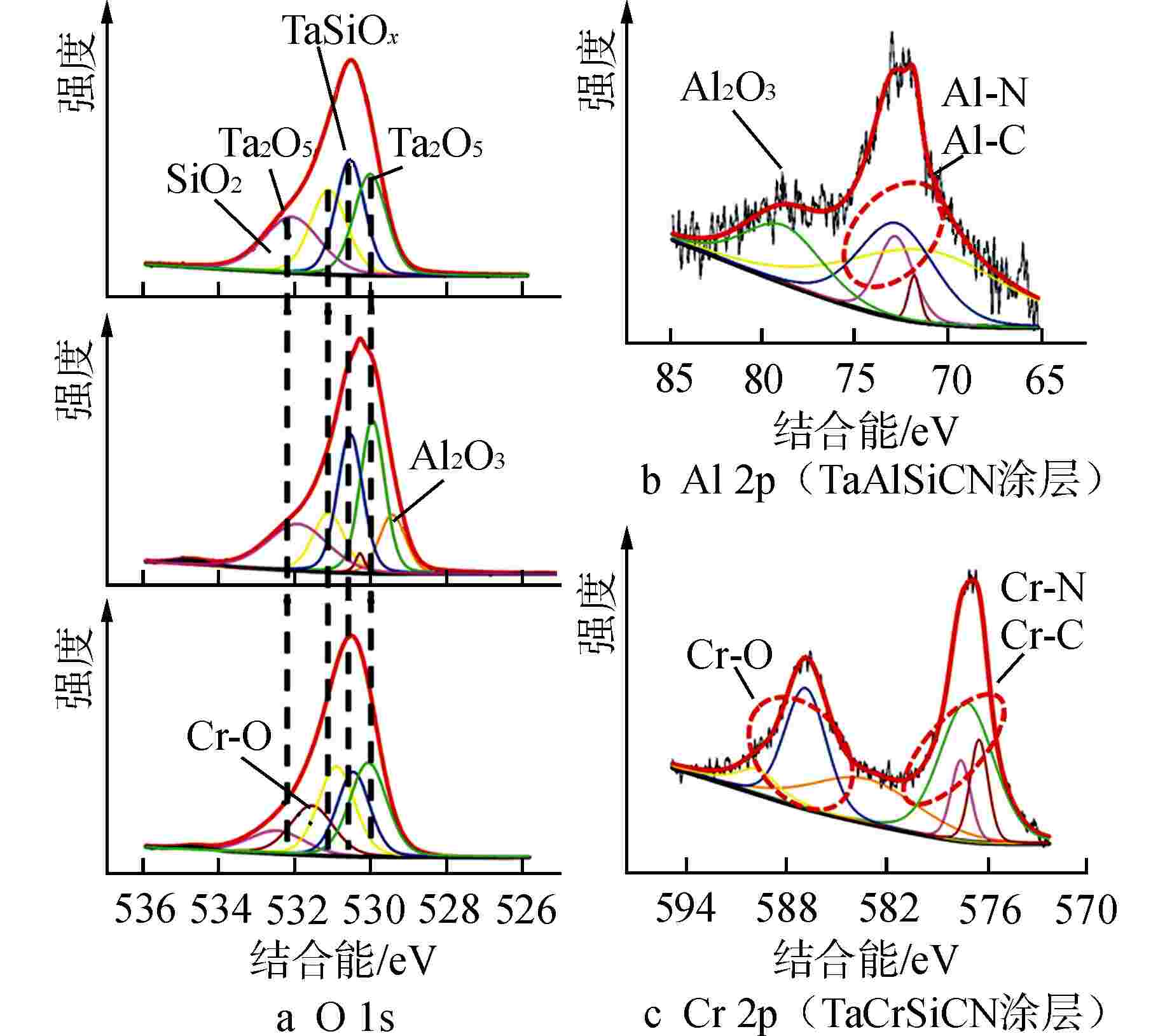
图 6 腐蚀4 h后涂层的XPS图谱
图6a能级谱图中从上到下依次为TaSiCN、TaAlSiCN、TaCrSiCN涂层
Figure 6. XPS Core Level Spectra of Coatings after 4 h Corrosion
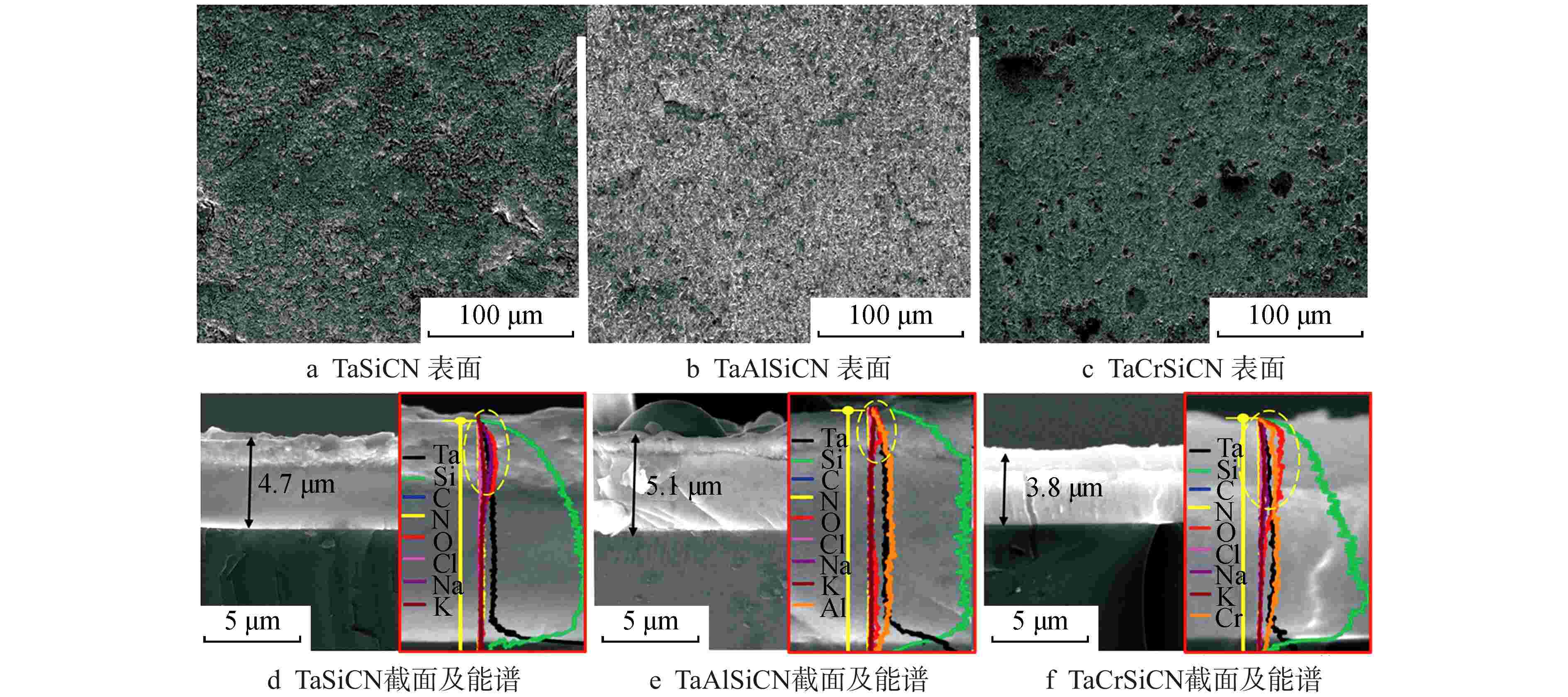
表 1 TaSiCN、TaAlSiCN和TaCrSiCN涂层的EDS元素分析
Table 1. EDS Elemental Composition of TaSiCN, TaAlSiCN and TaCrSiCN Coatings
试样 气体流量/(mL·min−1) 元素原子百分比/% Ar N2 Ta Si C N Al Cr TaSiCN 48 6 26 6 40 29 0 0 TaAlSiCN 48 6 22 8 34 26 10 0 TaCrSiCN 48 6 21 9 33 24 0 13 表 2 TaSiCN、TaAlSiCN和TaCrSiCN涂层表面和截面内部的EDS元素分析
Table 2. EDS Element Analysis of Surface and Cross Section of TaSiCN, TaAlSiCN and TaCrSiCN Coatings
分析区域 元素原子百分比/% Ta Si C N O Al Cr Cl Na K 涂层表面 TaSiCN 18.1 4.4 20.7 4.3 44.2 0.6 6.8 0.9 TaAlSiCN 12.8 4.8 28.2 6.6 40.5 3.2 0.5 6.8 1.4 TaCrSiCN 17.9 4.9 25.5 13.3 24.7 9.6 0.1 3.8 0.2 涂层截面内部 TaSiCN 19.9 8.5 25.8 38.4 5.6 0 1.8 0 TaAlSiCN 21.6 6.3 26.1 32.2 3.8 9.8 0 0.2 0 TaCrSiCN 17.9 7.9 23.1 31.2 7.2 10.3 0 2.4 0 -
[1] CHEN L, WANG J, ZONG Z H, et al. A new rock mass classification system QHLW for high-level radioactive waste disposal[J]. Engineering Geology, 2015, 190: 33-51. doi: 10.1016/j.enggeo.2015.02.006 [2] TANABE H, INAGAKI Y. The state of the art for high level radioactive waste (HLW) disposal in various countries[J]. Soil Mechanics & Foundation Engineering, 1998, 46: 15-18. [3] CRISTALDI M, IERADI L A, LICASTRO E, et al. Environmental impact of nuclear power plants on wild rodents[J]. Acta Zoologica Fennica, 1985, 173: 205-207. [4] SHANKAR A R, THYAGARAJAN K, MUDALI U K. Corrosion behavior of candidate materials in molten LiCl-KCl salt under argon atmosphere[J]. Corrosion, 2013, 69(7): 655-665. doi: 10.5006/0746 [5] NING Z E, LUO X F, ZHANG W, et al. Corrosion resistance of TaSiCN coatings in NaCl-KCl molten salt[J]. Materials Research Express, 2019, 6(10): 106437. doi: 10.1088/2053-1591/ab3ed6 [6] KUPTSOV K A, KIRYUKHANTSEV-KORNEEV P V, SHEVEYKO A N, et al. Comparative study of electrochemical and impact wear behavior of TiCN, TiSiCN, TiCrSiCN, and TiAlSiCN coatings[J]. Surface and Coatings Technology, 2013, 216: 273-281. doi: 10.1016/j.surfcoat.2012.11.058 [7] MANULYK A. Oxidation resistance mechanism of TiAlSiCN and TiCrSiCN compositions made by plasma spark sintering at 1200℃[M]//SINGH M, OHJI T, DONG S, et al. Advances in High Temperature Ceramic Matrix Compo Sites and Materials for Sustainable Development; Ceramic Transactions, Volume CCLXIII. The American Ceramic Society, 2017: 341-351. [8] SHTANSKY D V, KUPTSOV K A, KIRYUKHANTSEV-KORNEEV P V, et al. Comparative investigation of Al- and Cr-doped TiSiCN coatings[J]. Surface and Coatings Technology, 2011, 205(19): 4640-4648. doi: 10.1016/j.surfcoat.2011.04.012 [9] SHTANSKY D V, LEVASHOV E A, SHEVEIKO A N, et al. The structure and properties of Ti–B–N, Ti–Si–B–N, Ti–Si–C–N, and Ti–Al–C–N coatings deposited by magnetron sputtering using composite targets produced by self-propagating high-temperature synthesis (SHS)[J]. Journal of Materials Synthesis and Processing, 1998, 6(1): 61-72. doi: 10.1023/A:1022663210694 [10] CÓRDOBA J M, CHICARDI E, POYATO R, et al. Spark plasma sintering of Ti x Ta1− x C0.5N0.5-based cermets: effects of processing conditions on chemistry, microstructure and mechanical properties[J]. Chemical Engineering Journal, 2013, 230: 558-566. doi: 10.1016/j.cej.2013.06.104 [11] XIE Z W, WANG L P, WANG X F, et al. Influence of high temperature annealing on the structure, hardness and tribological properties of diamond-like carbon and TiAlSiCN nanocomposite coatings[J]. Applied Surface Science, 2011, 258(3): 1206-1211. doi: 10.1016/j.apsusc.2011.09.072 [12] ABD EL-RAHMAN A M, WEI R H. Effect of ion bombardment on structural, mechanical, erosion and corrosion properties of Ti–Si–C–N nanocomposite coatings[J]. Surface and Coatings Technology, 2014, 258: 320-328. doi: 10.1016/j.surfcoat.2014.09.006 [13] SHTANSKY D V, KUPTSOV K A, KIRYUKHANTSEV-KORNEEV P V, et al. High thermal stability of TiAlSiCN coatings with “comb” like nanocomposite structure[J]. Surface and Coatings Technology, 2012, 206(23): 4840-4849. doi: 10.1016/j.surfcoat.2012.05.068 [14] MANULYK A. MAX phases: understanding of erosion, corrosion and oxidation resistance properties in TiAlSiCN and TiCrSiCN compositions[J]. MRS Online Proceedings Library, 2016, 1812(1): 9-15. [15] XU H, NIE X, WEI R. Tribological behavior of a TiSiCN coating tested in air and coolant[J]. Surface and Coatings Technology, 2006, 201(7): 4236-4241. doi: 10.1016/j.surfcoat.2006.08.066 [16] WANG Y, LI J L, DANG C Q, et al. Influence of carbon contents on the structure and tribocorrosion properties of TiSiCN coatings on Ti6Al4V[J]. Tribology International, 2017, 109: 285-296. doi: 10.1016/j.triboint.2017.01.002 [17] LIN J L, WEI R H, GE F F, et al. TiSiCN and TiAlVSiCN nanocomposite coatings deposited from Ti and Ti-6Al-4V targets[J]. Surface and Coatings Technology, 2018, 336: 106-116. doi: 10.1016/j.surfcoat.2017.10.009 [18] VEPREK S, VEPREK-HEIJMAN M G J. The formation and role of interfaces in superhard nc-MenN/a-Si3N4 nanocomposites[J]. Surface and Coatings Technology, 2007, 201(13): 6064-6070. doi: 10.1016/j.surfcoat.2006.08.112 [19] VEPREK S, ZHANG R F, VEPREK-HEIJMAN M G J, et al. Superhard nanocomposites: origin of hardness enhancement, properties and applications[J]. Surface and Coatings Technology, 2010, 204(12-13): 1898-1906. doi: 10.1016/j.surfcoat.2009.09.033 [20] VEPREK S, VEPREK-HEIJMAN M G J, KARVANKOVA P, et al. Different approaches to superhard coatings and nanocomposites[J]. Thin Solid Films, 2005, 476(1): 1-29. doi: 10.1016/j.tsf.2004.10.053 [21] PETROV I, MYERS A, GREENE J E, et al. Mass and energy resolved detection of ions and neutral sputtered species incident at the substrate during reactive magnetron sputtering of Ti in mixed Ar+N2 mixtures[J]. Journal of Vacuum Science & Technology A, 1994, 12(5): 2846-2854. [22] WEI R, LANGA E, RINCON C, et al. Deposition of thick nitrides and carbonitrides for sand erosion protection[J]. Surface and Coatings Technology, 2006, 201(7): 4453-4459. doi: 10.1016/j.surfcoat.2006.08.091 [23] STANDARDS N I O. NIST X-ray Photoelectron Spectroscopy Database[Z]. 2001. [24] CHOI S R, PARK I W, KIM S H, et al. Effects of bias voltage and temperature on mechanical properties of Ti–Si–N coatings deposited by a hybrid system of arc ion plating and sputtering techniques[J]. Thin Solid Films, 2004, 447-448: 371-376. doi: 10.1016/S0040-6090(03)01085-X [25] SCHOCK M R. Response of lead solubility to dissolved carbonate in drinking water[J]. Journal AWWA, 1980, 72(12): 695-704. doi: 10.1002/j.1551-8833.1980.tb04616.x -





 下载:
下载: