Study on Weight-gain Model of FeCrAl Alloy by Steam Oxidation at Medium and High Temperature
-
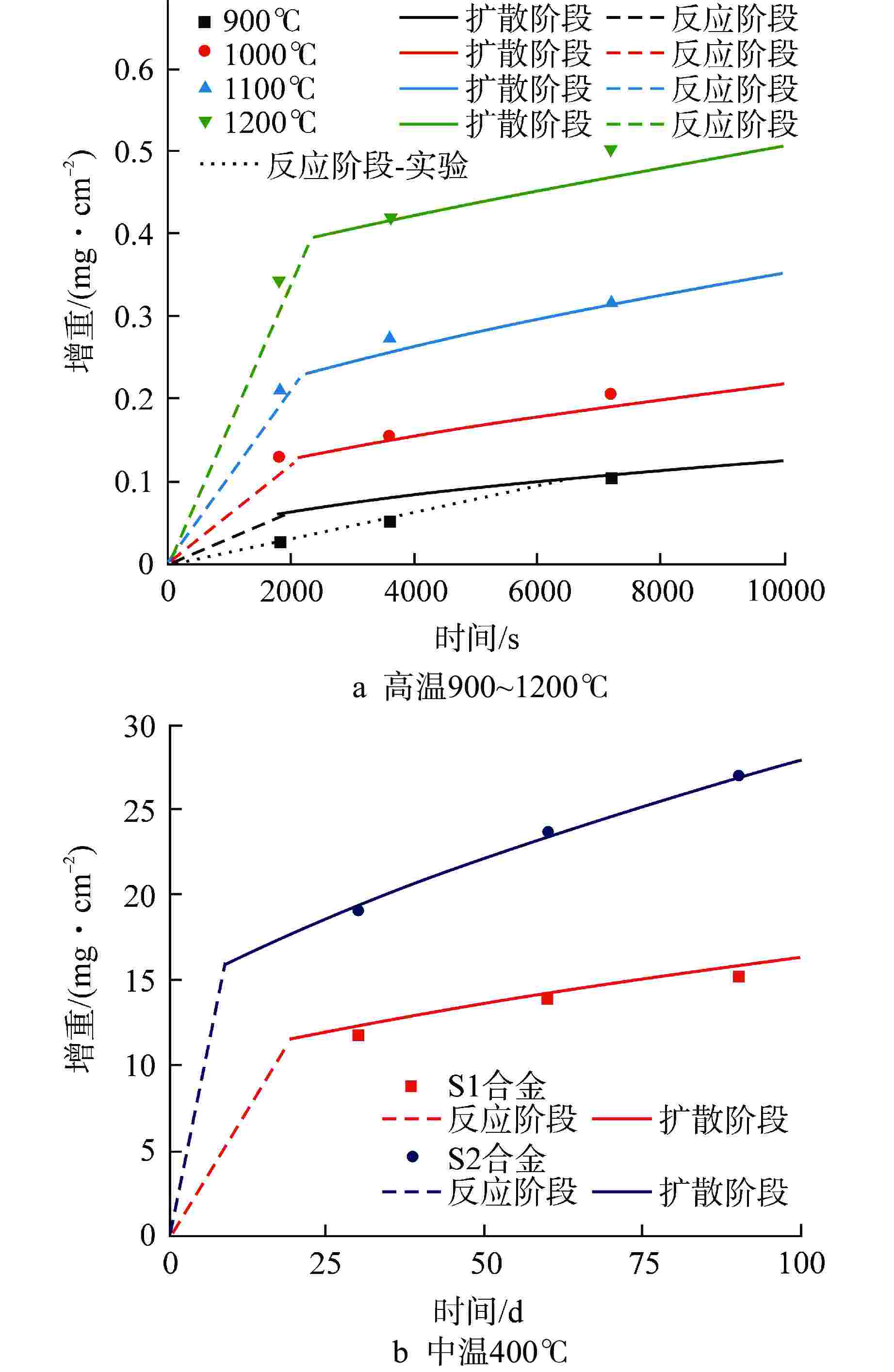
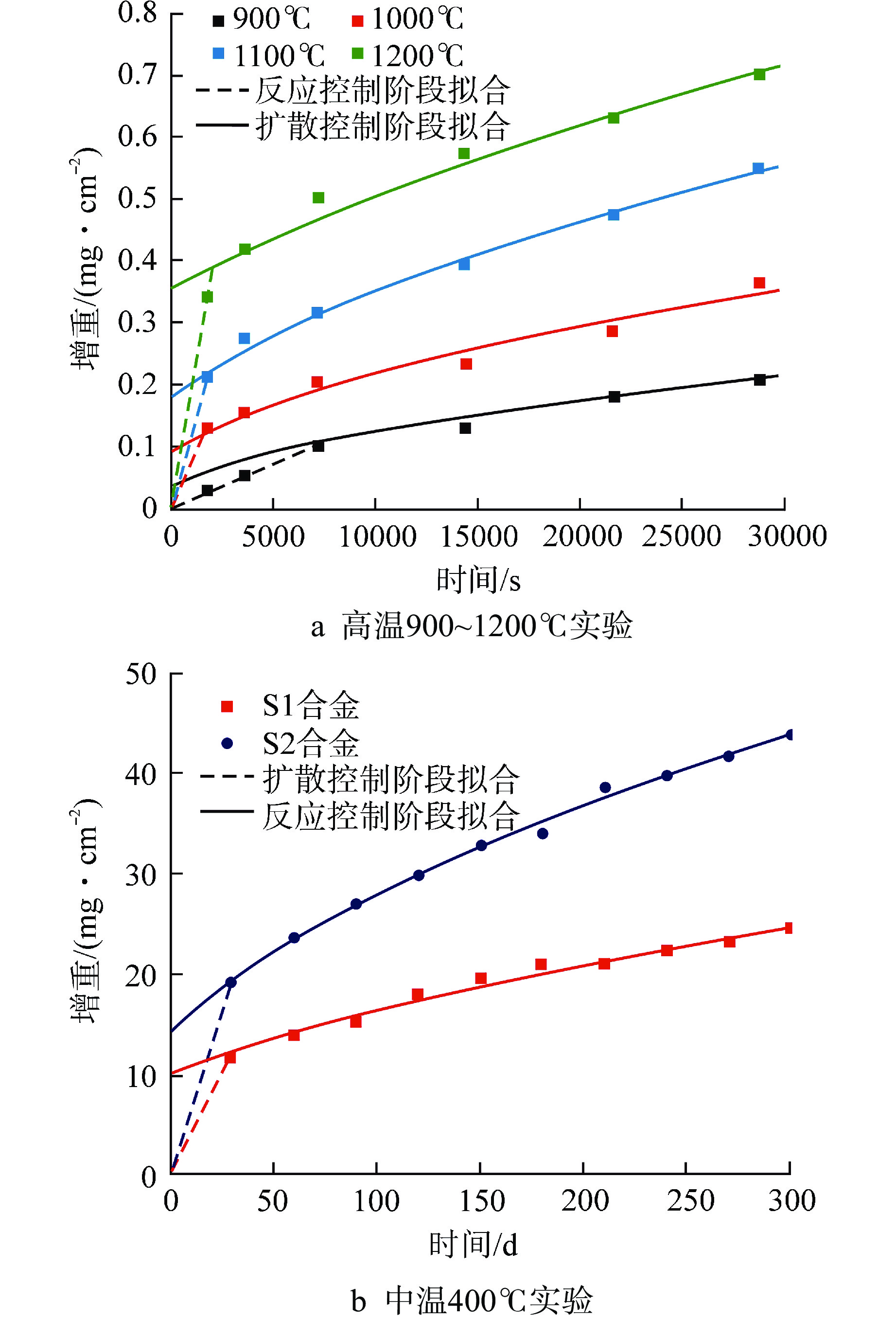
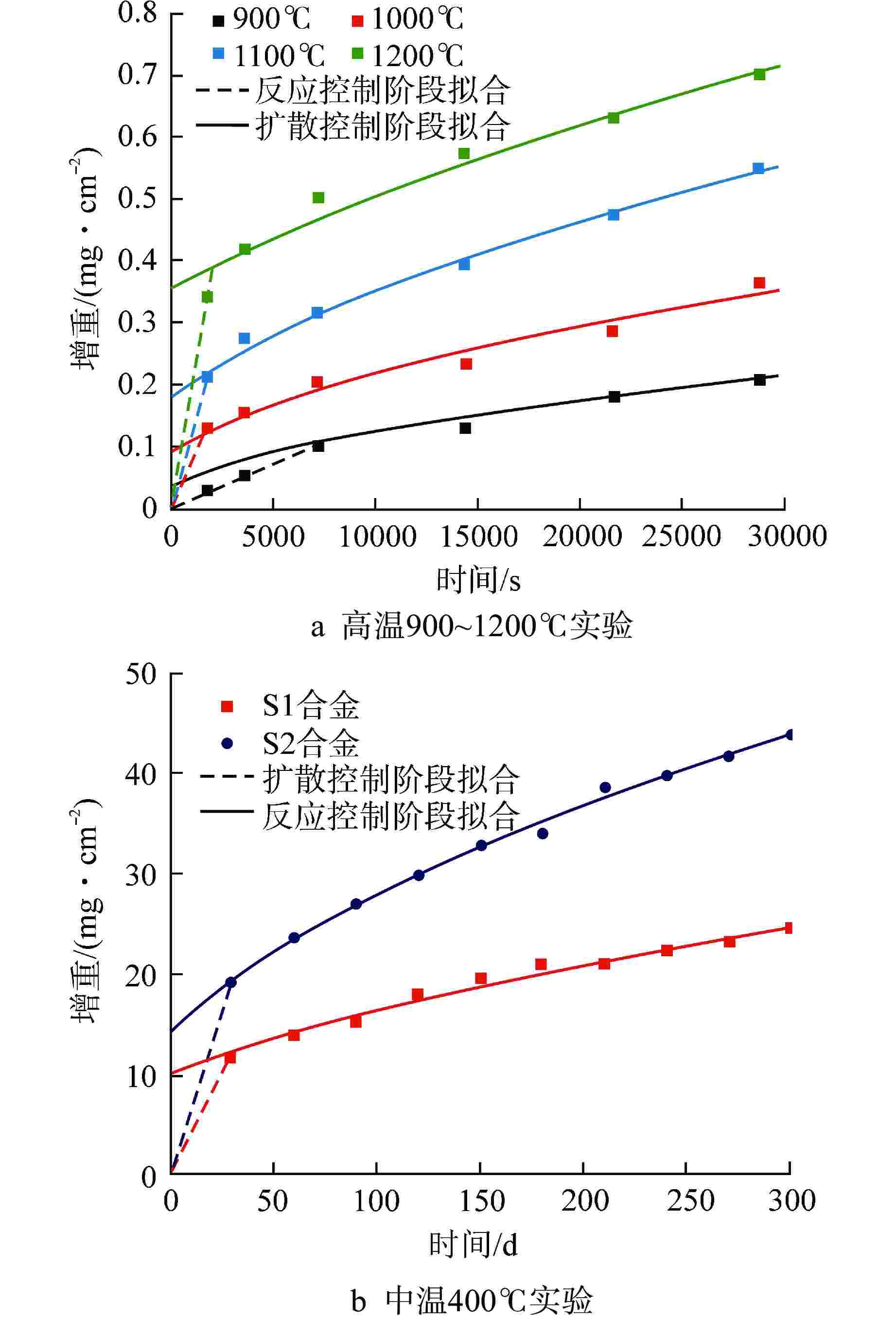
摘要: 通过预测FeCrAl合金在不同温度下的水蒸气氧化行为,从而为反应堆失水事故(LOCA)下的FeCrAl包壳性能演化仿真提供模型。本文基于反应控制与扩散控制的氧化机制,提出了两段式的氧化增重微分模型,并给出了模型参数标定方法。结合实验提供的FeCrAl合金在高温(900~1200℃)与中温(400℃)条件下的水蒸气氧化增重数据,模型能够统一描述400~1200℃温度区间内的FeCrAl合金氧化增重行为,与实验数据的误差控制在20%以内。同时观测到,反应-扩散机制的临界增重在400~900℃时基本不变,在更高温度时显著上升,其原因是高温时氧化层生长过快,难以形成致密的氧化保护层。考虑实际LOCA时初始水腐蚀氧化层的影响以及气压变化,模型给出了相对应的修正方案。本研究有望为FeCrAl合金包壳在LOCA下的失效行为仿真提供氧化增重模型与数据。
-
关键词:
- FeCrAl合金 /
- 耐事故燃料(ATF) /
- 氧化增重 /
- 高温水蒸气氧化 /
- 失水事故(LOCA)
Abstract: In order to predict the steam oxidation behavior of FeCrAl alloy at different temperatures and provide the model for the evolution simulation of the performance of FeCrAl cladding under loss of coolant accident (LOCA), a two-stage differential oxidation weight-gain model was proposed based on the reaction and diffusion control mechanisms, and a parameter calibration method was also presented. Combined with the experimental data from FeCrAl steam oxidation tests at high temperature (900-1200℃) and medium temperature (400℃), the model can uniformly describe the weight-gain behavior of FeCrAl alloy in the temperature range of 400-1200℃, and the error with experimental data is controlled within 20%. At the same time, it is observed that the critical weight-gain of the reaction-diffusion mechanism is basically unchanged at 400-900℃, but increases significantly at higher temperature, because the oxidation layer grows too fast to form the dense oxidation protective layer. In addition, considering the influence of initial oxide layer from water corrosion and the change of steam pressure during LOCA, a modified scheme of the oxidation-weight gain model is given. This study is expected to provide oxidation model and parameters for the failure behavior simulation of the FeCrAl alloy cladding under LOCA accidents. -
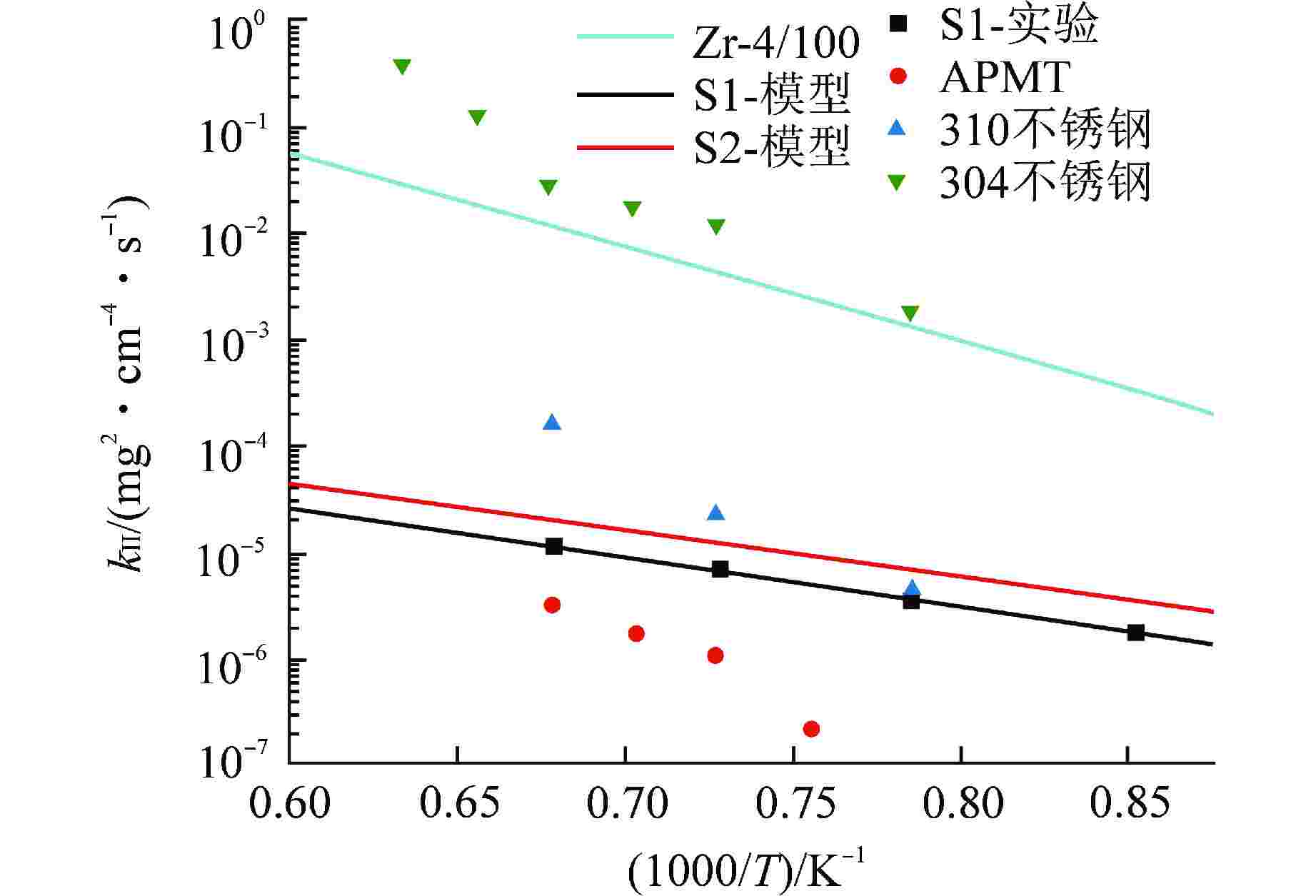
图 6 各类合金在高温水蒸气扩散阶段的氧化速率比较[3]
Figure 6. Comparison of High Temperature Steam Oxidation Rate at Diffusion Stage for Different Types of Alloys
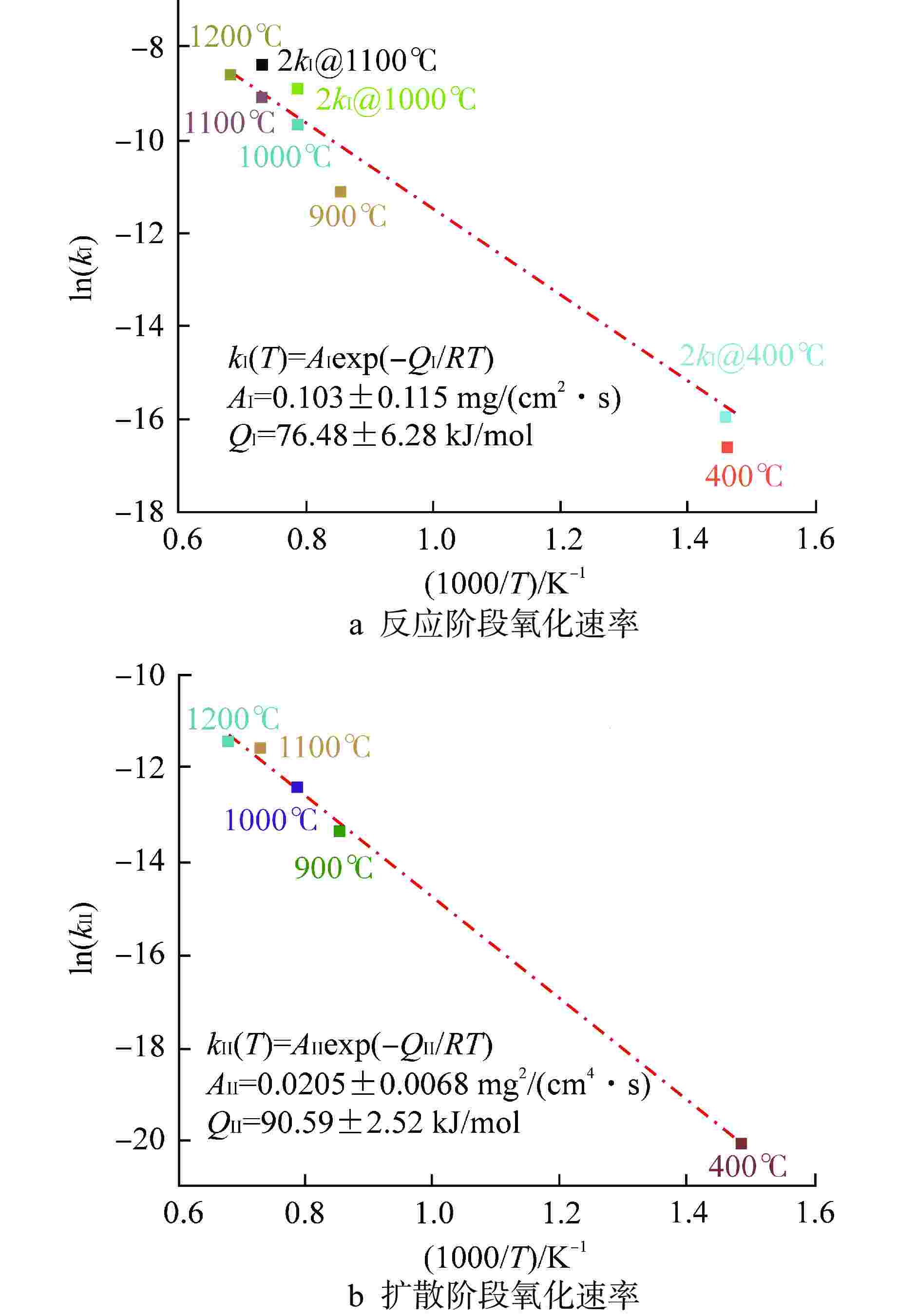
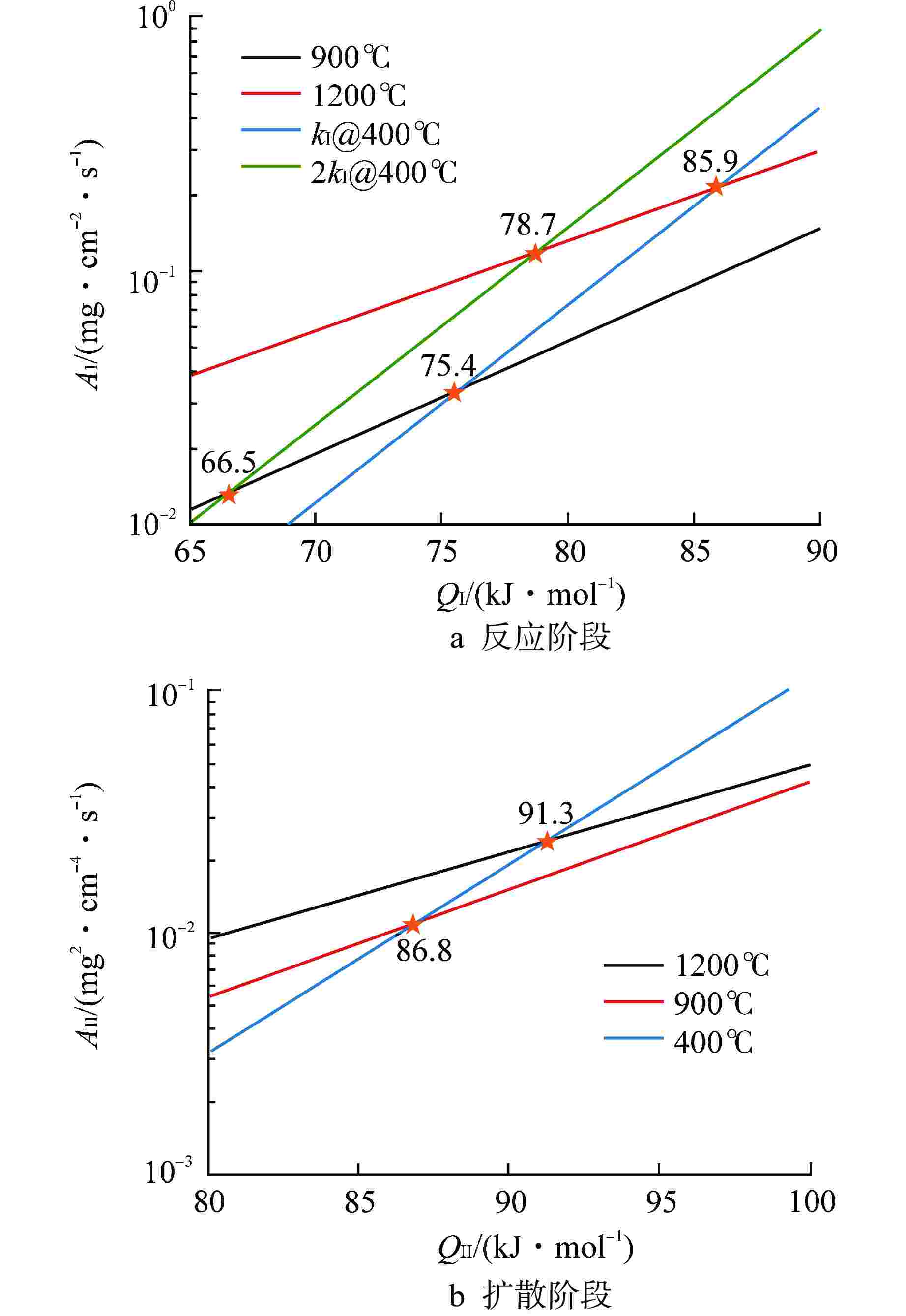
表 1 FeCrAl合金水蒸气氧化增重模型参数
Table 1. Parameters of FeCrAl Alloy Oxidation Weight Gain Model
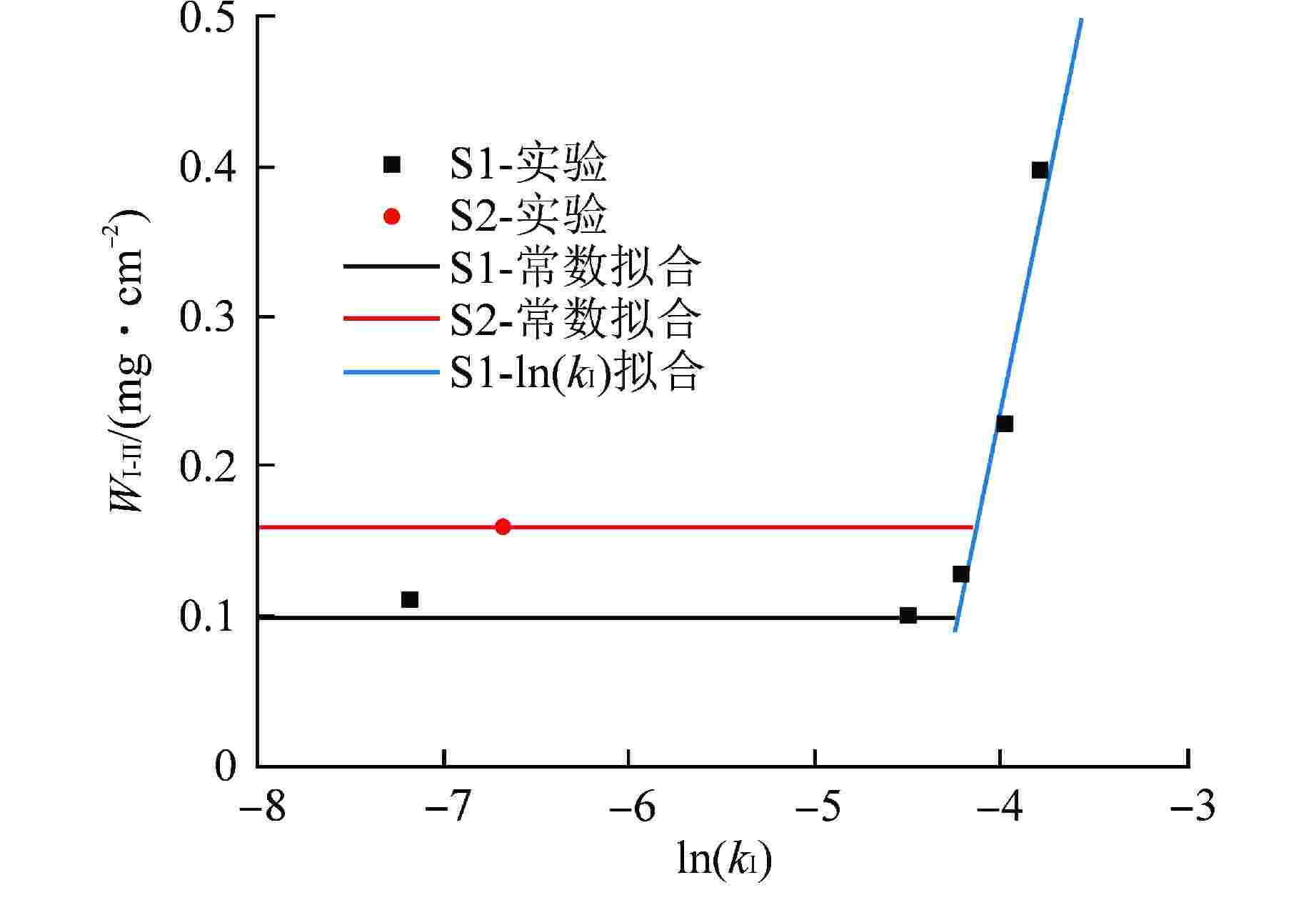
样品 T/℃ $ {Q_{\text{I}}} $/(kJ·mol–1) $ {A_{\text{I}}} $/(mg·cm–2·s–1) $ {Q_{{\text{II}}}} $/(kJ·mol–1) $ A\mathrm{_{II}} $/(mg2·cm–4·s–1) $ W_{\text{I-II}} $/(mg·cm–2) S1合金 900 80.4 0.12 89.1 0.016 0.06(0.10) 1000 0.13 1100 0.23 1200 0.40 400 0.11 S2合金 400 74.2 0.12 82.2 0.016 0.16 括号内数据指模型修正后的数据 -
[1] ALLEN T, BUSBY J, MEYER M, et al. Materials challenges for nuclear systems[J]. Materials Today, 2010, 13(12): 14-23. doi: 10.1016/S1369-7021(10)70220-0 [2] AZEVEDO C R F. Selection of fuel cladding material for nuclear fission reactors[J]. Engineering Failure Analysis, 2011, 18(8): 1943-1962. doi: 10.1016/j.engfailanal.2011.06.010 [3] TERRANI K A, ZINKLE S J, SNEAD L L. Advanced oxidation-resistant iron-based alloys for LWR fuel cladding[J]. Journal of Nuclear Materials, 2014, 448(1-3): 420-435. doi: 10.1016/j.jnucmat.2013.06.041 [4] ZINKLE S J, TERRANI K A, GEHIN J C, et al. Accident tolerant fuels for LWRs: a perspective[J]. Journal of Nuclear Materials, 2014, 448(1-3): 374-379. doi: 10.1016/j.jnucmat.2013.12.005 [5] TERRANI K A. Accident tolerant fuel cladding development: promise, status, and challenges[J]. Journal of Nuclear Materials, 2018, 501: 13-30. doi: 10.1016/j.jnucmat.2017.12.043 [6] WU X, KOZLOWSKI T, HALES J D. Neutronics and fuel performance evaluation of accident tolerant FeCrAl cladding under normal operation conditions[J]. Annals of Nuclear Energy, 2015, 85: 763-775. doi: 10.1016/j.anucene.2015.06.032 [7] UNOCIC K A, HOELZER D T, PINT B A. Microstructure and environmental resistance of low Cr ODS FeCrAl[J]. Materials at High Temperatures, 2015, 32(1-2): 123-132. doi: 10.1179/0960340914Z.00000000088 [8] JÖNSSON B, LU Q, CHANDRASEKARAN D, et al. Oxidation and creep limited lifetime of kanthal APMT®, a dispersion strengthened FeCrAlMo alloy designed for strength and oxidation resistance at high temperatures[J]. Oxidation of Metals, 2013, 79(1-2): 29-39. doi: 10.1007/s11085-012-9324-4 [9] PINT B A, TERRANI K A, YAMAMOTO Y, et al. Material selection for accident tolerant fuel cladding[J]. Metallurgical and Materials Transactions E, 2015, 2(3): 190-196. doi: 10.1007/s40553-015-0056-7 [10] TERENTYEV D, HAFEZ HAGHIGHAT S M, SCHÄUBLIN R. Strengthening due to Cr-rich precipitates in Fe–Cr alloys: effect of temperature and precipitate composition[J]. Journal of Applied Physics, 2010, 107(6): 061806. doi: 10.1063/1.3340522 [11] FIELD K G, HU X X, LITTRELL K C, et al. Radiation tolerance of neutron-irradiated model Fe–Cr–Al alloys[J]. Journal of Nuclear Materials, 2015, 465: 746-755. doi: 10.1016/j.jnucmat.2015.06.023 [12] YAMAMOTO Y, PINT B A, TERRANI K A, et al. Development and property evaluation of nuclear grade wrought FeCrAl fuel cladding for light water reactors[J]. Journal of Nuclear Materials, 2015, 467: 703-716. doi: 10.1016/j.jnucmat.2015.10.019 [13] SUN Z Q, BEI H B, YAMAMOTO Y. Microstructural control of FeCrAl alloys using Mo and Nb additions[J]. Materials Characterization, 2017, 132: 126-131. doi: 10.1016/j.matchar.2017.08.008 [14] SUN Z Q, YAMAMOTO Y. Processability evaluation of a Mo-containing FeCrAl alloy for seamless thin-wall tube fabrication[J]. Materials Science and Engineering:A, 2017, 700: 554-561. doi: 10.1016/j.msea.2017.06.036 [15] BADINI C, LAURELLA F. Oxidation of FeCrAl alloy: influence of temperature and atmosphere on scale growth rate and mechanism[J]. Surface and Coatings Technology, 2001, 135(2-3): 291-298. doi: 10.1016/S0257-8972(00)00989-0 [16] WANG P, QI W, YANG K, et al. Systematic investigation of the oxidation behavior of Fe-Cr-Al cladding alloys in high-temperature steam[J]. Corrosion Science, 2022, 207: 110595. doi: 10.1016/j.corsci.2022.110595 [17] PINT B A, DRYEPONDT S, UNOCIC K A, et al. Development of ODS FeCrAl for compatibility in fusion and fission energy applications[J]. JOM, 2014, 66(12): 2458-2466. doi: 10.1007/s11837-014-1200-z [18] CHENG T, KEISER J R, BRADY M P, et al. Oxidation of fuel cladding candidate materials in steam environments at high temperature and pressure[J]. Journal of Nuclear Materials, 2012, 427(1-3): 396-400. doi: 10.1016/j.jnucmat.2012.05.007 -





 下载:
下载: